The Space Surrounding The Nucleus Of An Atom Contains
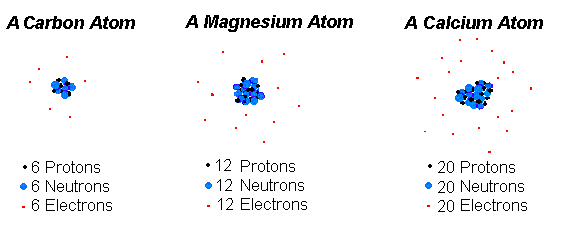
In fact for all practical purposes the mass of the atom is the sum of the masses of the protons and neutrons.
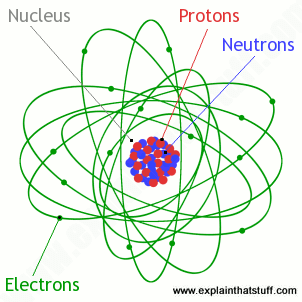
The space surrounding the nucleus of an atom contains. The nucleus contains neutrons and protons. An electron is an elementary particle that is a fundamental constituent of matter having a negative charge and existing independently or as the component outside the nucleus of an atom. What are found in the space surrounding the nucleus of an atom. Good luck to you.
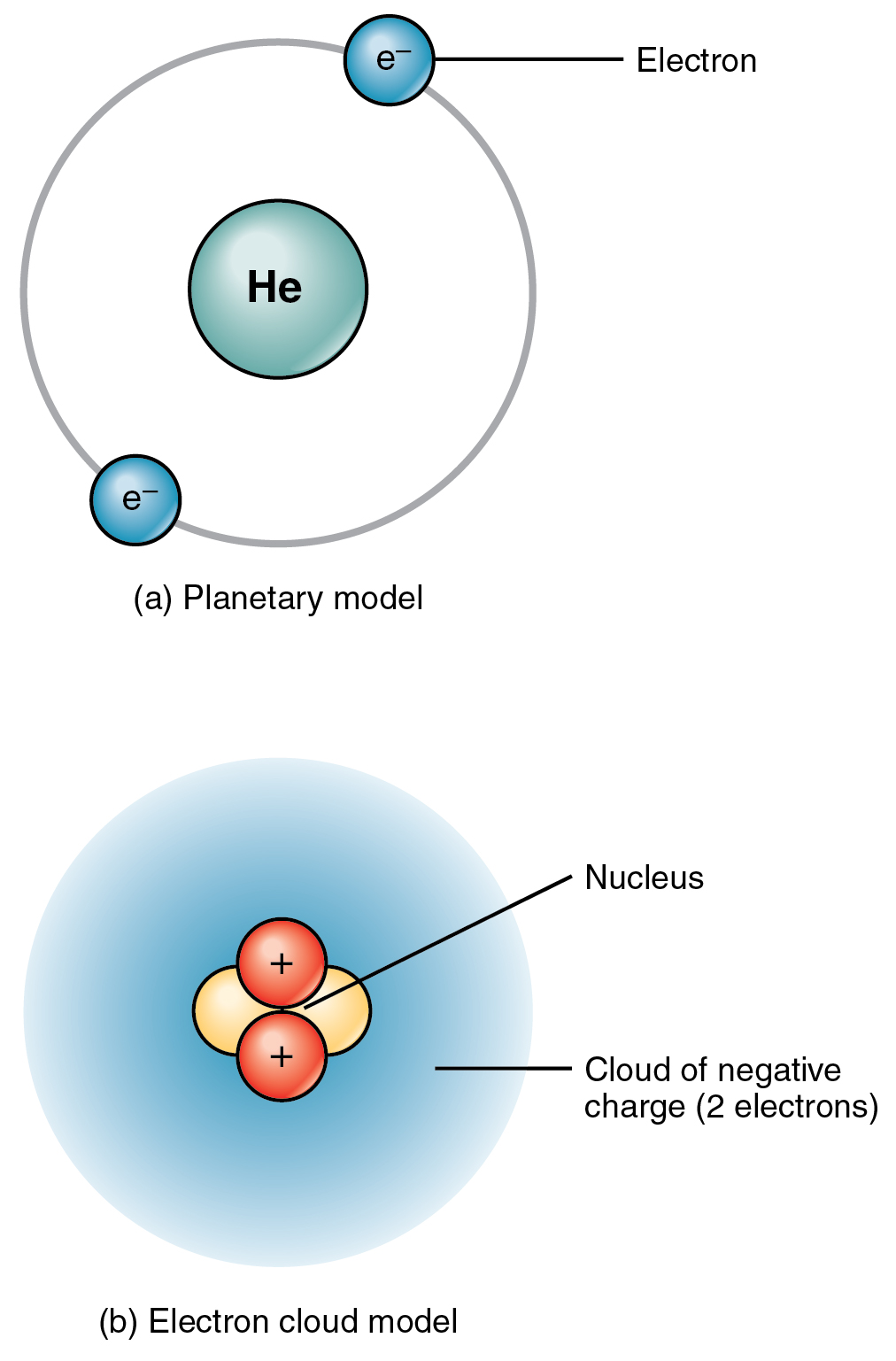
The diameter of the atom is about 100 000 times bigger than the diameter of the nucleus. The space surrounding the nucleus of an atom contains a. The protons of an atom are all crammed together inside the nucleus. In the electron clouds surrounding the nucleus are electrons.
If an atom contains 3 protons 4 neutrons and 3 electrons its mass number is. Electrons move in orbits around the nucleus. The protons and neutrons form a very small dense core known as the nucleus. Isotopes are tons of the same element with the same number of protons and a different number or.
The nucleus is very very small and very very dense when compared to the rest of the atom. The nucleus contains the protons which carry a positive charge. The space surrounding the nucleus of an atom contains a. Protons neutrons and electrons.
Therefore most of the mass of an atom is contained in its nucleus. So your answer is b electrons. Each proton carries a positive charge and like charges repel each other.